
Organ transplants have revived the dead so it’s not all that different to the classic gothic tale of Frankenstein by Mary Shelley. My fascination for the tale as well as the tissue engineering and stem-cell lectures led to a deep dive into this topic. The tale depicts Doctor Frankenstein’s creation of life from post-mortem organs that haunt him, but now, centuries later in modern medicine, it suggests a futuristic prophecy.
Creating Life in the Lab
Regenerative medicine is still in its early phases but there has been tremendous advancement. Pluripotent stem cells are cells that can differentiate into any type of cell in the body, so work has been conducted to grow organs from scratch with these cells. However, it’s difficult to replicate the intricate tissue organisation and complexities of organs. Just like Doctor Victor Frankenstein, scientists are itching to discover the secrets of “life and death” and learn how to “renew life”.
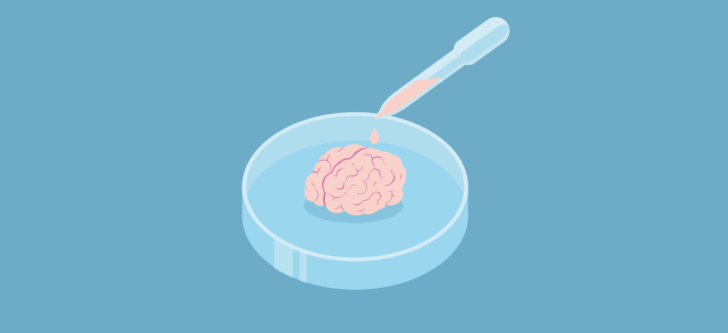
Although not applicable for transplantation, miniature organoids (‘organ-like’) have been created around various laboratories to replicate hearts, kidneys and more. Biomedical scientists such as myself can use organoids as models when testing a new drug, without harming patients. It can help understand and assess drug safety before a new drug is given to humans in clinical trials to save a lot of money and time spent in trials. In the Netherlands these organoids were used to personalise medicine for cystic fibrosis patients who were treated based on how an organoid grown from their cells behaved in response to drugs.
Organ Transplant
Transplantation of human organs is regarded as one of the key markers of 20th-century medicine. The first successful kidney transplant was performed in 1954 and since then the demand for transplant has skyrocketed. Thus, doctors have attempted to put artificial and animal-derived organs into patients. On the one hand scientific curiosity such as this has advanced human knowledge and life expectancy, but it makes me wonder at what point can curiosity go too far?
Ethics
I always wondered how medical professionals could pass judgement and determine who is worthy of a transplant. I found that in the UK strict criteria exist, where some criteria are objective (e.g. blood type) while others depend on ethical judgement. It raises questions like:
-
- Should people’s lifestyle choices (smoking, obesity, drug use) that implicated their organs be given an opportunity for organ transplant?
- Should prisoners receive a transplant?
- Should people get a chance to get a second transplant?

David Bennett was the world’s first recipient of genetically modified pig heart after being denied a human heart. Media suggested the denial was due to his advanced heart failure while his son declared it was due to his non-compliance in managing his health. This debate continued after the surgery as news about his conviction after stabbing a man in the 1980s resurfaced. The unfortunate victim’s sister argued she wishes the modified heart “had gone to a deserving recipient.”
Final Thoughts
I think Frankenstein’s key message is that curiosity and ambition need to be met with caution and compassion, which I think has also shaped current ethical guidelines. Researching the complex interplay of science and ethics of organ transplant shifted my perspective into a more comprehensive and holistic one.







Very well written, with an excellent format and images. You’ve included interesting statistics and related it to personal ideas which…
This is a very well written blog, the format is as if you are talking directly to me. The ideas…
Love the Batman GIF :)
This is an excellent, well written blog. The narrative is engaging and easy to follow. It could be improved by…
This is a well-communicated blog. The it is written well with good use of multimedia. It could be improved with…